|
In contrast, locking is governed by rugged free energy landscape and is associated with the formation of β-sheets by A β peptides on the edges of amyloid fibrils. The former occurs without detectable free energy barriers and resembles polymer adsorption on attractive walls. 22 Our studies have suggested that the docking and locking stages are fundamentally different. Using computer simulations we have probed the thermodynamics of A β fibril growth by computing its free energy landscape. During the second (locking) stage a bound A β monomer adopts an ordered fibril conformation through structural reorganization. 18 – 21 During the first (docking) stage disordered incoming A β peptide binds to the fibril without integration into the fibril structure. 8, 9 Elongation of A β fibrils was proposed to proceed via addition of monomers according to the two-stage “dock-lock” mechanism. This structure reveals that A β peptides are organized into parallel in-registry β-sheets laminated into four layers. 17 Among them is a two-fold symmetry structure of A β 1–40 fibril, which is derived from the solid-state NMR experiments under agitated conditions 9 ( Fig. The most common A β species are 40-mer A β 1–40 fragments, which have been shown to form polymorphic amyloid fibrils. 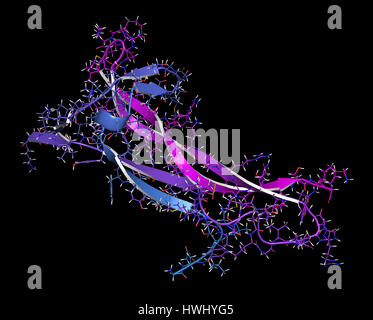
14 – 16Ī β peptides, the fragments of amyloid precursor protein cleaved during cellular proteolysis, are the amyloidogenic species, which are linked to the onset of Alzheimer’s disease. 13 In contrast to structurally ordered fibrils, oligomers sample multitude of conformations and exhibit a distribution of sizes starting with dimers. 7 – 12 Backbone hydrogen bonds (HBs) linking polypeptide chains into β-sheets and side chain interactions render amyloid fibrils remarkably stable against dissociation. Structural studies have uncovered a remarkable homogeneity of amyloid fibril cores based on β-sheet structure. 4 Biomedical studies have showed that oligomeric species 5 and, to a lesser extent, fibrils 6 have cytotoxic properties. 1 – 3 Amyloid formation is linked to more than 20 various disorders, including Alzheimer’s, Parkinson’s, and Creutzfeldt-Jakob diseases. 1 Amyloid formation is a complex multistage conformational transition, which is initiated with the oligomerization of monomers and progresses with the development of protofibrils and mature amyloid fibrils as final products. Polypeptide chains show apparent generic propensity to assemble into supramolecular ordered structures called amyloid fibrils. These simulation findings together with the backbone mutagenesis experiments suggest that a viable strategy for arresting fibril growth is the disruption of interpeptide HBs. As for the peptides bound to A β fibril the deletion of interpeptide HBs reduces the β propensity in the dimers making them less competent for amyloid assembly. As a result the deletion of peptide-fibril HBs is expected to impede fibril growth. Without the support of HBs bound A β peptides form few long β-strands on the fibril edge. 
However, cancellation of HBs forming between incoming A β peptides and the fibril disrupts the locked fibril-like states in the bound peptides. Their elimination does not change the continuous character of A β binding nor its temperature. 
The HBs also do not play a critical role in initial binding of A β peptides to the amyloid fibril. The backbone HBs have little effect on the stability of A β dimers or on their aggregation interface. The importance of HBs appears to depend on the aggregation stage. Using REMD and united atom implicit solvent model we examine the role of backbone hydrogen bonds (HBs) in A β aggregation.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
